#Pfizer seeks COVID vaccine authorization for 12-15 year olds in US
“#Pfizer seeks COVID vaccine authorization for 12-15 year olds in US”
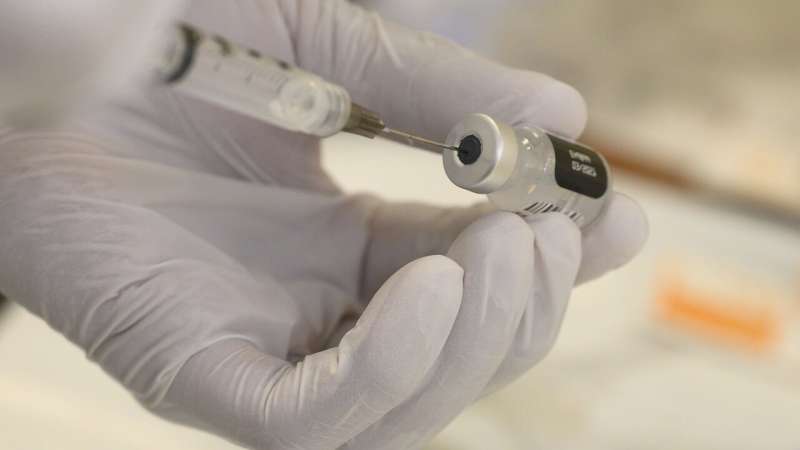
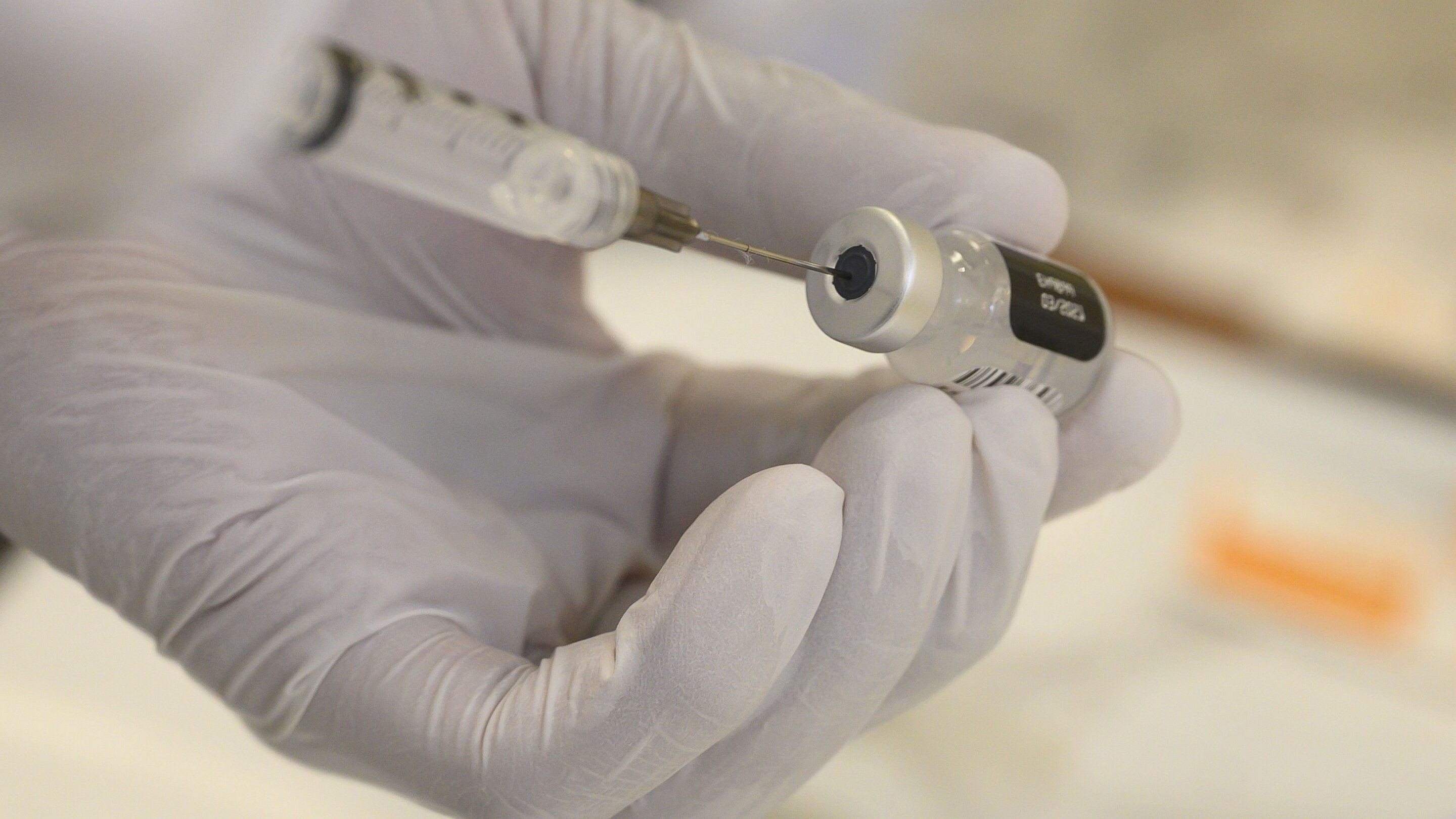
Pfizer-BioNTech asked for authorization Friday to use their COVID-19 vaccine on 12-15 year olds in the United States.
The companies said in a statement that they plan to make similar requests of other regulatory authorities worldwide in coming days.
Their request to the Food and Drug Administration in the US comes after Phase 3 clinical trials of the Pfizer vaccine in 12-15 year olds were 100 pc effective in warding off the disease, according to the companies.
In late March they published the results of trials carried out with 2,260 adolescents in the US who the companies said showed “robust anti-body responses.”
The vaccine was “well tolerated with side effects generally consistent with those observed in participants 16 to 25 years of age,” the companies said Friday.
For now the vaccine has emergency authorization for use in people 16 and older.
Children are less likely to develop severe COVID so vaccinating them has been less of a priority than getting older people inoculated.
The BioNTech/Pfizer shot is based on novel mRNA technology and was the first COVID-19 vaccine to be approved in the West late last year.
Follow the latest news on the coronavirus (COVID-19) outbreak
© 2021 AFP
Citation:
Pfizer seeks COVID vaccine authorization for 12-15 year olds in US (2021, April 9)
retrieved 9 April 2021
from https://medicalxpress.com/news/2021-04-pfizer-covid-vaccine-authorization-.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.
If you liked the article, do not forget to share it with your friends. Follow us on Google News too, click on the star and choose us from your favorites.
For forums sites go to Forum.BuradaBiliyorum.Com
If you want to read more Like this articles, you can visit our Science category.