#Instrument at BESSY II shows how light activates molybdenum disulfide layers to become catalysts
“#Instrument at BESSY II shows how light activates molybdenum disulfide layers to become catalysts”
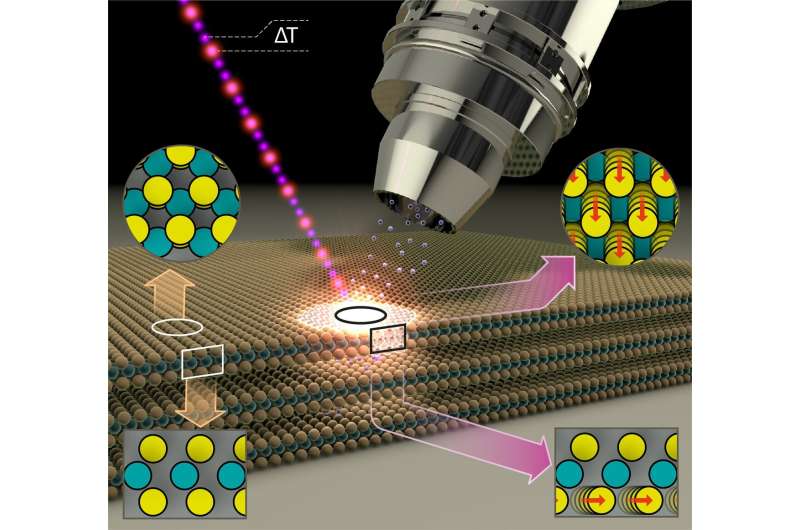
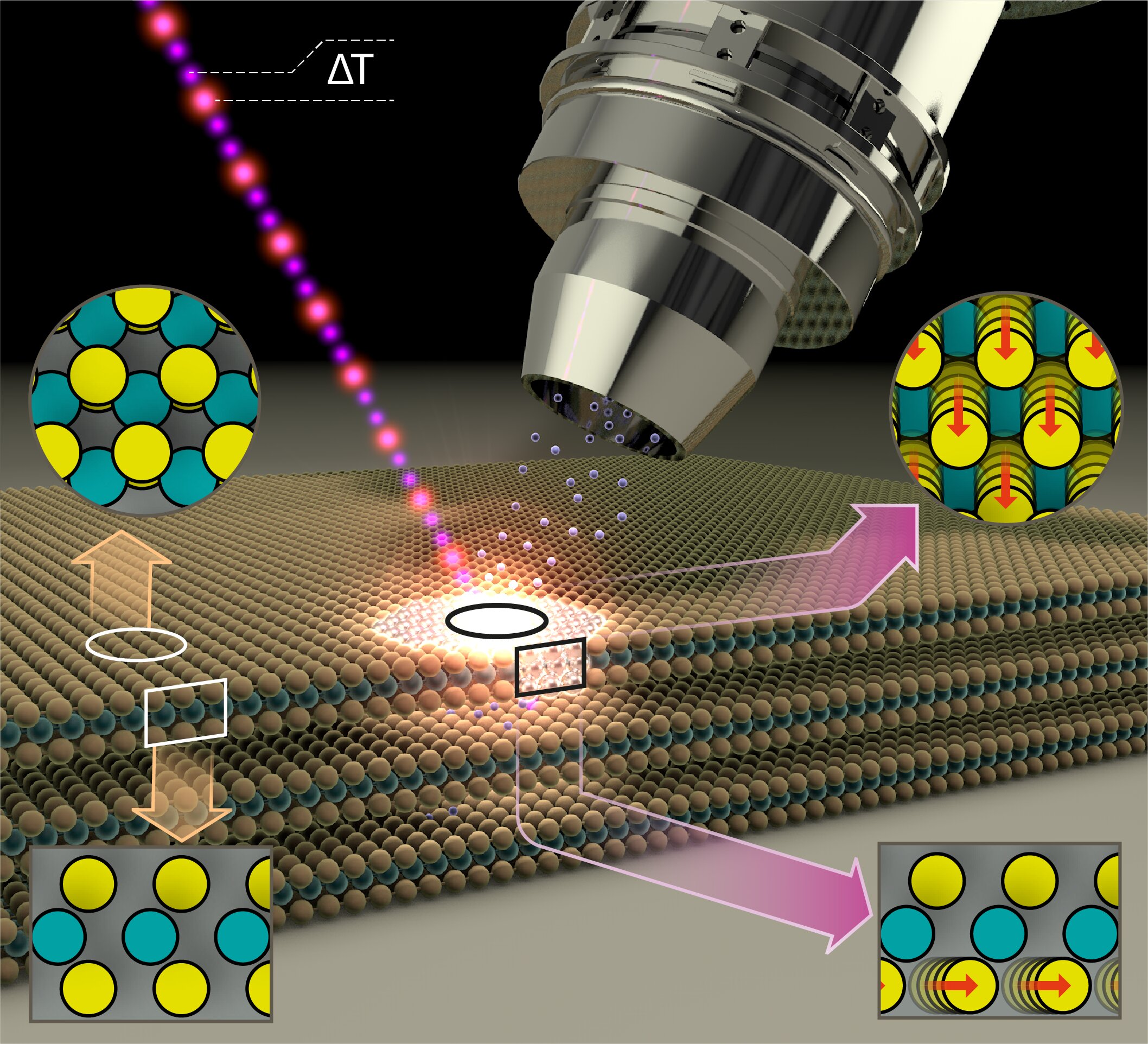
MoS2 thin films of superposed alternating layers of molybdenum and sulfur atoms form a two-dimensional semiconducting surface. However, even a surprisingly low-intensity blue light pulse is enough to alter the properties of the surface and make it metallic. This has now been demonstrated by a team at BESSY II.
The exciting thing is that the MoS2 layers in this metallic phase are also particularly active catalytically. They can then be employed, for example, as catalysts for splitting of water into hydrogen and oxygen. As inexpensive catalysts, they could facilitate the production of hydrogen—an energy carrier whose combustion produces no CO2, only water.
Physicist Dr. Nomi Sorgenfrei and her team have constructed a new instrument at BESSY II to precisely measure the changes in samples using temporally-resolved electron spectroscopy for chemical analysis (trESCA) when irradiating the samples with low-intensity, ultra-short light pulses. These light pulses are generated at BESSY II using femtosecond time-slicing (femtoslicing) and are therefore both low intensity and extremely short duration. The new instrument, named SurfaceDynamics@FemtoSpeX, can also rapidly obtain meaningful measurements of electron energies, surface chemistry, and transient alterations using these low-intensity light pulses.
Analysis of the empirical data showed that the light pulse leads to a transient accumulation of charge at the surface of the sample, triggering the phase transition at the surface from a semiconducting to a metallic state.
“This phenomenon should also occur in other representatives of this class of materials, the p-doped semiconducting dichalcogenides, so it opens up possibilities of influencing functionality and catalytic activity in a deliberate way,” Sorgenfrei explains.
Phasing out a microscope’s tricks
Nomi L. A. N. Sorgenfrei et al, Photodriven Transient Picosecond Top‐Layer Semiconductor to Metal Phase‐Transition in p‐Doped Molybdenum Disulfide, Advanced Materials (2021). DOI: 10.1002/adma.202006957
Citation:
Instrument at BESSY II shows how light activates molybdenum disulfide layers to become catalysts (2021, March 5)
retrieved 6 March 2021
from https://phys.org/news/2021-03-instrument-bessy-ii-molybdenum-disulfide.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.
If you liked the article, do not forget to share it with your friends. Follow us on Google News too, click on the star and choose us from your favorites.
For forums sites go to Forum.BuradaBiliyorum.Com
If you want to read more Like this articles, you can visit our Science category.



